The Unfair Stigmatization of Vitamin A
Share
The Unfair Stigmatization of Vitamin A by Pam Schoenfield
8/11/2016
The Unfair Stigma
If you take a prenatal vitamin, look on the label to see whether it contains vitamin A (some brands actually don’t contain any vitamin A), it will probably indicate “from beta-carotene or mixed carotenoids.” When you see this, think carrots, sweet potatoes or papaya, all excellent sources of the pro-vitamin A precursor beta-carotene. You may recall hearing “eat your carrots—they are good for your eyes.” Indeed, for many people, eating orange fruits and vegetables supplies beta-carotene, which can be converted to retinal for the light-absorbing pigments in the retina.
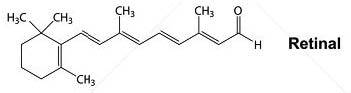
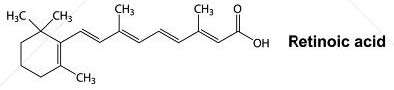
Vitamin A can take one of three forms in the human body—retinol, retinal and retinoic acid—depending on its level of oxidation and purpose. Normal embryonic development depends upon retinoic acid for the correct expression of the receptors in the differentiating cells—that is, for the transition from undifferentiated stem cells into differentiated cells such as heart cells, bone cells and brain cells. For this differentiation to occur, vitamin A supplied by the diet must be available in adequate forms and quantities.
While essential throughout the lifespan for growth, immunity, epithelial tissue maintenance, lung and visual function, “[vitamin A’s] influence is particularly critical during periods when cells proliferate rapidly and differentiate, such as during pregnancy and early childhood.”1 Deficiencies of vitamin A produce a myriad of deleterious outcomes in animals including defects in the eyes, snout, dental arches and in the worst case, spontaneous abortion or death of mother and offspring during labor, as described by Weston A. Price.2 In humans, even mild deficiencies during pregnancy can lead to compromised kidney development in the child.3
Vitamin A’s role in reproduction has been solidly established. But questions remain. What is the optimal amount of vitamin A during pregnancy? Are women getting enough to support the health of their babies? Is it possible to get too much?
RETINOL, RETINAL AND RETINOIC ACID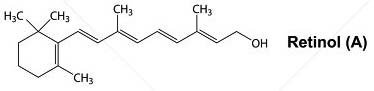
Recommended Dietary Intakes VERSUS Actual Population Intakes
The National Academy of Sciences Institute of Medicine (IOM) has established the recommended dietary allowance (RDA)4of vitamin A for non-pregnant females at 700 μg/day of retinol activity equivalents (RAE, explained below), and the RDA for pregnant females at 770 μg/day RAE (2570 IU). This is only 70 μg or just 10 percent more than the requirement for a non-pregnant female. The questionable methodology used to determine the pregnancy RDA has been discussed by Chris Masterjohn, PhD.3 For newborn infants, an adequate intake is 400 μg/day (1320 IU)—more than half the total RDA during pregnancy.5 Current public health guidelines advise that people consume at least 2.5 cups of fruits and vegetables a day to ensure adequate nutrient intake of RAE from carotenoids. However, the U.S. Centers for Disease Control and Prevention (CDC) also states: “fish-liver oils, liver, egg yolks, butter, and cream are known for their higher content of [preformed] vitamin A.”6
The IOM set the tolerable upper intake level during pregnancy at 3,000 μg/day RAE (10,000 IU).7 The upper level only applies to the intake of preformed vitamin A (retinol) not to carotenoids, which are not assigned an upper level. Although beta-carotene is not limited during pregnancy, a high intake can cause carotenemia, which has been associated with amenorrhea in young vegetarian women.8 Recent research also suggests that cleavage products of beta-carotene can block vitamin A at its receptor sites—another possible anti-nutrient?9
According to USDA data, 53 percent of women ages eighteen to thirty have a usual intake of vitamin A from diet that falls below the estimated average requirement (EAR) of 500 μg RAE/day.10 An EAR is calculated to meet the requirements of 50 percent of the individuals in a gender/age group; half of this group will not have their needs met from intakes equal to the EAR. The RDA is set higher in order to include 97.5 percent of the individuals in the entire gender/age group. In the case of vitamin A, 53 percent of women in their prime reproductive years do not even meet this very low dietary intake of 500 μg (1670 IU). The usual dietary intakes in the lowest fifth and tenth percentiles of females ages eighteen to thirty are 210 μg and 257 μg respectively, only one-third or less of the RDA.11,12 The USDA named vitamin A one of the “shortfall nutrients,” but did not go a step further and designate it as one of the “nutrients of concern” which would indicate a substantial public health risk.13
The FDA provides guidance to manufacturers regarding vitamin supplement labeling: the FDA’s “daily value” of vitamin A for pregnant and lactating women continues to be 8,000 IU, since publication in the Federal Register in 1993.14 Recently, the FDA removed vitamin A from its mandatory Nutrition Facts Label for packaged foods, a move they made because they claim “[i]n the early 1990s, American diets lacked Vitamins A and C, but now Vitamin A and C deficiencies in the general population are rare.”15 It is unclear how the U.S. diet has changed to contain more vitamin A. However, because “vitamin A” on a label does not usually indicate the presence of true vitamin A, but rather carotenoids, this omission may not matter.
According to the CDC, more than 95 percent of the U.S. population has adequate serum vitamin A levels of 20 μg/dL or greater, in line with the World Health Organization (WHO) standard of less than 20 μg/dL used to define public health problems. The CDC has estimated the prevalence of low serum vitamin A among females to be 0.2 percent, or 317,000 American women.16 Yet the CDC acknowledges that: “[s]erum retinol values do not always reflect total body status because of homeostatic control and therefore are often not useful for assessing the vitamin A status of individuals. Additional tests may be required to confirm vitamin A deficiency when 20 μg/dL is used as a cutoff.”
According to a WHO report, in the U.S. vitamin A status can be poor, especially among low-income populations. In children qualifying for the Special Supplemental Nutrition Program for Women, Infants, and Children (WIC), 32 percent were in the “uncertain area” for the modified relative dose response (MRDR) values (an indication of low vitamin A stores). Furthermore, an assessment of low-income pregnant women showed that an alarming 9 percent were above the international MRDR cut-off of 0.060 for vitamin A adequacy (frankly vitamin A deficient).17 The MRDR cut-off is a more accurate method of determining vitamin A status, as explained below.
While stating that “high-dose” vitamin A should be avoided in pregnancy, particularly between day fifteen and day sixty following conception, the WHO recommends vitamin A supplementation in regions where deficiency is common for the prevention of night blindness. Supplementation of 10,000 IU/day or 25,000 IU/week is recommended for pregnant women in these areas, beginning after day sixty of gestation and continuing for at least twelve weeks or until delivery.18 (Note that “high dose” vitamin A of 50,000 to 400,000 IU has typically been administered within six to eight weeks postpartum in areas of endemic vitamin A deficiency, with the higher amount being more effective at raising breast milk concentrations.19) Perhaps it would be wise to consider the real probability that many “well-nourished” American women are living in a country where vitamin A deficiency is common?
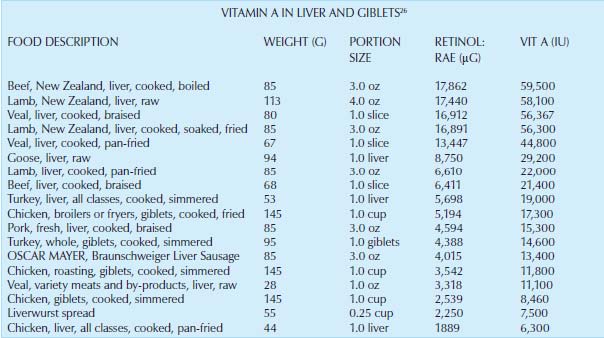
LIVER FOR VITAMIN A
In 2015, the European Food Safety Authority set population reference intakes at 650 μg (2170 IU) for non-pregnant women and 700 μg (2333 IU) during pregnancy. The German Nutrition Society recommends a 40 percent increase in vitamin A intake for pregnant women equal to 1100 μg RAE (3670 IU).20 Unfortunately, “pregnant women or those considering becoming pregnant are generally advised to avoid the intake of vitamin A-rich liver and liver foods, based upon unsupported scientific findings. As a result, the provitamin A carotenoid β-carotene remains their essential source of vitamin A.”21
“The average intake of β-carotene in Germany is about 1.5–2 mg a day. . . the total vitamin A contribution from β-carotene intake represents 10–15 percent of the RDA.”21 Without sources of retinol, it is highly unlikely German women will meet these recommendations. Compared to other foods, “the only relevant dietary source for vitamin A is liver. All other foods containing preformed vitamin A need to be consumed in atypically large amounts in order to meet the vitamin A requirement. Only 10–15 g [1/2 ounce] of animal liver is necessary to meet the daily requirement, demonstrating that liver is the most important source of vitamin A for humans.”21 Other foods like meat, butter, eggs and milk only contribute a small portion (less than 20 percent) of dietary preformed vitamin A.21 We would agree that liver makes a unique contribution to the diet—that of easily satisfying vitamin A needs.
The UK and Australian guidelines state that evidence is lacking to support routine supplementation of vitamin A and that excessive quantities of fat-soluble vitamins may cause harm. They go further to recommend that pregnant women avoid consuming liver and liver products.22,23,24
While true that “excessive” quantities of fat-soluble vitamins can be harmful, does the practice of eating four ounces of liver weekly lead to an excessive intake of vitamin A? A reasoned examination of the evidence seems to say no. Also, what about the recent trend of vitamin D supplementation during pregnancy, sometimes as high as 5,000 IU/day; is supplementing this fat-soluble vitamin in isolation not without harm?25
At the second Hohenheim Nutrition Conference in Germany in 2009, Dr. Georg Lietz reported that a high percentage of women in the UK are at risk for vitamin A deficiency; one of the chief causes is the fact that two common genetic variations (one or both were present in almost half of healthy women studied) greatly lessen the body’s ability to convert beta-carotene into vitamin A. If a women carries both genes that reduce this conversion, she can have a one-third to two-third reduction in the ratio of retinyl palmitate (active vitamin A) to beta-carotene circulating in her blood.27 Studies conducted by scientists with the USDA Agricultural Research Service confirm that 45 percent of individuals cannot achieve adequate vitamin A status from beta-carotene alone.28,29
In addition, the absorption of beta-carotene and other carotenoids can be quite unpredictable—food factors and other individual differences can significantly affect bioavailability as well as the conversion to retinol. While 70-90 percent of preformed vitamin A (retinol) is absorbed, even under ideal conditions only 3 percent or less of dietary carotenoids are assimilated. Foods also vary widely in the amount of carotenoids they contain due to varietal differences, ripeness, climate during cultivation, storage, processing and preparation methods. For example, carrots cooked in fat will have more absorbable carotenoids than raw carrots, and more if puréed.30 Beta-carotene from supplements is much more readily absorbed than beta-carotene from food sources.
Dr. Lietz draws attention to a problem that is also common in the U.S.: the low consumption of vitamin A-rich foods. “Worryingly, younger women are at particular risk. The older generation tend to eat more eggs, milk and liver, which are naturally rich in vitamin A, whereas the health-conscious youngsters on low-fat diets are relying heavily on the beta-carotene form of the nutrient.”27 Of course this is now a common practice among elders, too.
Despite the likelihood that low intakes in women are much more common than high intakes, strong warnings to avoid preformed vitamin A during pregnancy from authoritative sources continue. According to the Embryo Project: “[e]xcess intake of vitamin A and retinoids by pregnant women often results [in] malformations to fetuses’ skulls, faces, limbs, eyes, central nervous system.” The use of “often” is erroneous and highly misleading, unless they are specifically referring to synthetic retinoids such as Accutane.31,32 Many other organizations and “experts” advise against eating liver, and any other food naturally high in vitamin A, such as cod liver oil.33 Typically, couples planning to start a family fail to question this advice.
A young Chinese expectant mother once told me, “I actually like to eat liver but not while I am pregnant because it is not good for the baby.” This is an unfortunate impression as liver was part of her family’s traditional foodways. Another patient hoping to become pregnant said, “You don’t want too much vitamin A in pregnancy because it can cause birth defects.” But is this true? What historically was the practice of eating liver while pregnant? Was there a good reason to discontinue this practice? What are the risks, if any, of consuming preformed vitamin A?
Liver and cod liver oil, both high in vitamin A, are traditional foods valued by many indigenous cultures.34 It wasn’t long ago that books advising mothers on what to eat during pregnancy placed liver on the top of the list, not only for vitamin A, but also vitamins B1, B2, B6, B12 and protein.35 And with good reason. Liver is an excellent source of all these vitamins as well as choline and cholesterol; all are needed for successful reproduction. Cod liver oil was a mainstay of the diet for many families prior to the 1970s. In 1941, the UK instituted the Vitamin Welfare Scheme, which provided cod liver oil in black currant syrup to improve the nutrition of vulnerable groups including expectant mothers.36
The Erie, New York, Department of Health publication “Eating Right During Pregnancy” places liver and liver sausage first under “Be sure to include these foods rich in iron in your diet.”37 What this health department may not recognize is the fact that vitamins A and B6 are also required to build red blood cells; “iron deficiency” anemia may never be completely resolved without supplying these two vitamins in adequate amounts.38,39 (B12 and folate are required for red blood cells as well.) The American Pregnancy Association offers similar advice: “The U.S. RDA recommends about 27 mg [iron] per day. Lean beef, chicken, lamb, liver, turkey and veal are good options.”40
Certainly not all nutrition experts advise against the consumption of foods or supplements high in preformed vitamin A. According to Dr. Micozzi: “In fact, a lack of vitamin A, especially during pregnancy and in infancy, poses far greater risks. Deficiency of vitamin A in developing babies is known to cause birth defects, poor tooth enamel, a weakened immune system, and literally several hundred thousand cases of blindness per year worldwide. This is why developing countries safely give megadoses of vitamin A to newborns.”41
Likewise, highly respected medical organizations, including the March of Dimes42 and the Mayo Clinic,43 do not list liver among the foods to limit or avoid completely during pregnancy. But rather than relying on expert opinions, let’s consider what “far greater risks” may present themselves if a women enters pregnancy with low vitamin A status.
Vitamin A deficiency during pregnancy is known to increase the risk of maternal mortality and is associated with premature birth, intrauterine growth retardation, low birthweight, and abruptio placentae. Ideally the prevention of birth defects and other poor outcomes will begin before conception. An important key to prevention is the mother and father consuming a fully nourishing traditional diet. All major organ systems develop early in pregnancy, often before a women knows that she is pregnant.44 Vitamin A signalling is essential during the development of several organs including eyes, brain, liver, kidneys and heart.45,46
A personal story may be useful here. Functional nutrition expert Chris Kresser responded to a question on vitamins for pregnancy via his blog.47 A comment was posted by a mother who believes a lack of vitamin A in her own diet was the cause of her daughter’s congenital kidney condition, vesicoureteral reflux (VUR). While no one can be sure of cause and effect, it is troubling that urinary tract defects are the most common class of defects in developed countries. According to this mother, “the pediatric urologist said he didn’t know why kids get VUR but that it was probably hereditary since a child with the disorder has a 30 percent chance of having a sibling with the disorder as well. I think he’s only halfway right—a mother who is malnourished for one pregnancy is likely to be malnourished for another, especially subsequent pregnancies from the first one where something went wrong developmentally.”
In support of this mother’s suspicion, a study of pregnant women with short birth intervals or multiple births showed that almost one-third of the women had plasma retinol levels below 1.4 μmol/l corresponding to a borderline deficiency.21 A short time period between pregnancies is a risk factor for a number of adverse outcomes including prematurity, low birth weight and neural tube defects. The leading hypothesis suggests that short intervals may be a marker for a deficiency in nutritional reserves needed to support normal fetal development. Maternal stores of critical micronutrients such as vitamins A, B6, B12, D3, zinc and folate, decline during pregnancy and may take many months to rebound.48
THE ROTHMAN STUDY
The evidence points to the very real problem of vitamin A insufficiency during pregnancy, an issue unlikely to be addressed by conventional medical authorities in the near future.1 It was the Rothman study49 that sealed the stigmatization against vitamin A during pregnancy; this study has been critically examined by Masterjohn who states that the preponderance of the evidence supports intakes of vitamin A between 20,000 and 25,000 IU during pregnancy to reduce the risk of birth defects.3
From the CDC’s response to the Rothman study: “Daily consumption of a single multivitamin preparation containing folic acid and no more than the U.S. recommended daily allowance (RDA) of vitamin A for pregnant women (8000 IU) is beneficial. . . In 1987 the Centers for Disease Control, the Teratology Society, and the Council for Responsible Nutrition independently published recommendations designed to reduce pregnant women’s exposure to high amounts of vitamin A from supplements.”
“These recommendations were made because teratogenesis was assumed to occur at some undetermined level above 8000 IU of vitamin A per day and because pregnant women in the United States do not appear to benefit from additional vitamin A. The recommendations included limiting vitamin A in prenatal multivitamin preparations to 5000 to 8000 IU and the vitamin A content of all multivitamin preparations to 10,000 IU; suggesting that women should not take 10,000 IU or more of vitamin A without consulting a physician; using beta-carotene rather than vitamin A in supplements; and specifying the amounts of retinol, retinyl esters, and beta carotene on supplement labels.”50
“[Rothman and team] report findings consistent with those of other studies suggesting that the consumption of less than 10,000 IU of vitamin A per day from vitamin supplements is safe.” Specifically addressing Rothman’s findings that greater than 10,000 IU is teratogenic: “[i]t would have been useful if Rothman . . . had presented more detailed data on the amounts consumed by the women who took 10,000 IU or more of vitamin A per day and on the birth defects in their infants. Without further knowledge of this sort, [more detailed data on the women with the highest level of vitamin A consumption, very likely much more than 25,000 IU per day] we do not recommend using the dose–response curve in the study by Rothman . . . for the purpose of advising pregnant women who have consumed more than the RDA of vitamin A about the specific risk of malformation in their offspring. We cannot make good estimates of the teratogenicity of this vitamin at higher consumption levels unless more data become available.”50
How did we go from recommendations in 1995 to limit intake to 8,000-10,000 IU and prenatal vitamins routinely containing 8,000 IU of retinol51, to today where many prenatal vitamins contain no preformed vitamin A (and often little or no beta-carotene), compounded by warnings against liver consumption? According to Dr. Dean Raffelock, “[the Rothman study] created a wave of fear in obstetricians and their pregnant patients that continues to this day. . . In fact, obstetricians are often the most clueless, which is why they often recommend the extremely poor quality prenatal vitamins that the drug companies make.”52
In 2014, Pennsylvania State University scientists stated: “[n]utrition experts know that vitamin A is necessary for prenatal growth and development. . . but the role of vitamin A remains unclear for the neonatal [newborn] period. Stores of vitamin A become depleted as the fetus reaches full term, to the point where newborns are nearly depleted of vitamin A. Neonates born in developing countries are likely to have even lower amounts of vitamin A in their bodies. [Newborns] are born with a low vitamin A level but a high demand for it.”53
WIDESPREAD DEFICIENCY
Since the fetal liver is only able to store a small amount of vitamin A, it is generally accepted that almost all babies are born with marginal vitamin A deficiency. “Even term infants are relatively deficient in vitamin A at the time of birth compared to older children.”54 Premature infants have even lower liver stores of vitamin A at birth and their blood concentrations of retinol often remain low throughout their first year, putting them at risk for eye, chronic lung and gastrointestinal diseases.55 Vitamin A supplementation in pre-term infants reduces mortality and oxygen requirement and is considered a promising intervention for bronchopulmonary dysplasia prevention, a lung impairment common to preemies.56
Other scientists have expressed their concerns. “The fetus begins accumulating vitamin A during the third trimester and needs several months of sufficient intake after birth to build up an adequate store. The composition of breast milk is influenced by the vitamin A status of the mother during the last trimester. “[T]he main consequence of a poor vitamin A supply during pregnancy is a low vitamin A status at birth and in the next few months.”1
The vitamin A content in the breast milk of mothers who deliver prematurely is often closer to mature breast milk (which comes in three to four weeks after delivery). Both contain one-tenth to one-half the amount of vitamin A in the earliest milk, colostrum. Is there a connection between a deficiency of vitamin A in a premature infant and the lower content of the mother’s milk—perhaps an indication that the mother herself was deficient in vitamin A?
These findings pose serious questions. The first is, why are newborns (even in developed countries) born with low or depleted stores of vitamin A? Secondly, since experts know vitamin A is necessary, why are there so many warnings against it and why don’t prenatal vitamins contain it to any degree? Finally, if the mother’s vitamin A intake has little effect on her newborn’s vitamin A levels, why do newborns in developing countries have even lower amounts in their bodies compared to the developed world? One surmises that the widespread prevalence of maternal vitamin A deficiency in these countries may be a primary reason. Although stores of retinol in the livers of human fetuses have been found to be lower than those of adults, significant correlations have been measured between maternal serum retinol, fetal liver retinol and fetal growth.57
In adults, vitamin A stores are used up daily, at the rate of about 0.5 percent per day. This could occur more rapidly for those with lower liver stores, which depend on dietary intake.58 Vitamin A depletion could be accelerated during times of higher demand, such as infection or periods of rapid growth, which is why children’s dietary requirements are almost the same as adults. With little or no preformed vitamin A intake during a full-term pregnancy of about two hundred seventy days, an almost total reduction in liver stores after two hundred days is theoretically possible. Does conventional obstetrical care ever recommend vitamin A for “healthy” women?
According to a medical nutrition textbook, “because of the potential teratogenic effects from exposure to excess preformed vitamin A in the first trimester, vitamin A supplementation in pregnant women who are not deficient is not recommended.” Yet, these same authors add “women with low vitamin A intake or reduced liver stores should increase their intake throughout pregnancy to ensure adequate stores are available for rapid fetal growth in late gestation.”22
ASSESSING VITAMIN A STATUS
Assessing vitamin A status is difficult.1 Measuring liver reserves using a biopsy is considered the gold standard but this procedure is highly invasive and thus very rarely used. Clinical signs like poor dark adaptation, xerophthalmia (dry eyes), hyperkeratosis (hard bumps on skin), or even acne in adult women can yield clues, but the absence of these signs cannot rule out deficiencies.59 Night blindness is not likely to occur until liver stores are dangerously low.
Assessing intake not only requires detailed dietary records over several days (especially since people don’t eat liver very often) but also information on whether a woman is a good or poor converter of beta-carotene into retinol. Liver stores can be estimated using the relative dose response test (RDR or MRDR): blood is drawn, a dose of vitamin A is given, then the change in serum retinol is measured five hours later; an increase of 20 percent or more usually indicates deficient liver stores of vitamin A. This test has been used for population assessment but appears to be uncommon in clinical settings.59
Measuring blood levels of vitamin A is the more common way of determining vitamin A status. In healthy adults, the plasma vitamin A concentration ranges from 20 to 80 μg/L.60 However, blood levels can remain normal even when liver, lungs and other organ stores are low. Blood levels drop below the normal level of 20 μg/L only after liver stores are exceedingly low, so marginal liver stores may not be detected. A result of 10 μg/L indicates a severe deficiency and depleted liver stores.61
To determine either deficiency or excess states, measure both serum retinol (or serum vitamin A) and serum retinyl esters (serum A palmitate); serum retinol should ideally be in the middle or upper half of the reference range and not less than 30 μg/L, and the serum retinyl esters should not be above 10 percent of the sum of the retinol and retinyl esters. Measuring fasting retinyl esters is done to check whether the capacity of the liver to store vitamin A has been exceeded, a rare but possible occurrence.62
Balance with Vitamins D and K2
Supplementing with vitamin D is a common practice among obstetricians, often without the benefit of blood testing to determine what level of supplementation, if any, is optimal. More astute practitioners know that supplementing with vitamin D, especially at levels of 5,000 IU per day (which I have seen in pregnant patients) is not advisable unless attention is paid to ensuring vitamin A intake is optimal. Too much vitamin D can deplete vitamin A. Likewise, intake of vitamins A and D require balance with vitamin K2. Poultry liver, which contains all three fat-soluble vitamins—A, D and K2—is an ideal source of these synergistic nutrients.
Other considerations
Iron and zinc deficiency can affect the conversion to vitamin A. Iron is needed for the intestinal conversion of carotenoids to retinal; zinc is required for the subsequent conversion from retinal to retinol. Iron deficiency is not uncommon during pregnancy, and zinc status may be marginal especially in women with more vegetarian-type diets or higher needs.63 Zinc deficiency can exacerbate low vitamin A intakes because zinc is needed in the synthesis and secretion of retinol binding protein, which mobilizes vitamin A from the liver and transports it into circulation.64 Liver is an excellent source of both nutrients.
Iron deficiency anemia during pregnancy is typically treated with higher doses of iron. Yet increased intakes of vitamins A and B6 may be required to resolve the anemia, with or without supplemental iron depending on need. Vitamin A deficiency seems to impair mobilization of iron from body stores; therefore vitamin A supplementation improves hemoglobin concentration.64Serum retinol levels below 30 mcg/dL have been associated with “vitamin A-responsive” anemia. Since blood levels of retinol-binding protein change with pregnancy, the serum retinol test can yield inaccurate results.22 Be aware that routine iron supplementation is not advisable during pregnancy, since an increased iron intake in women with an already adequate iron status may have adverse effects. Assessment of serum ferritin at the onset of pregnancy will help determine whether more iron or iron-rich foods is indicated.
Conclusion
Clearly, vitamin A plays a vital role in pregnancy nutrition and the development of a healthy baby. It has been called “the concertmaster of fetal development”65 because it is needed to “achieve harmonious tissue organization.”1
If still uncertain whether consuming additional preformed vitamin A would be beneficial (either from cod or fish liver oil, other types of supplements, or from foods like liver), discuss this with your obstetrical care provider. In any case, 3000 μg RAE (10,000 IU) per day from all sources during pregnancy appears to be well within a safe upper limit according to most data; the Weston A. Price Foundation recommends at least 20,000 IU during pregnancy from foods such as liver or high vitamin cod liver oil along with 2,000 IU vitamin D (if sun exposure is limited) and vitamin K2 from aged cheese, animal
fats and fermented foods.
If you choose to avoid this or any amount of preformed vitamin A during pregnancy, it is advisable to build up your own liver stores by consuming more vitamin A-rich foods during the six to twelve months prior to conception. A woman considering pregnancy or already pregnant can also discuss her vitamin A needs with her obstetrical care provider and see whether further evaluation is warranted.22 Keep in mind that most obstetricians will not be aware of the controversy surrounding the limits on vitamin A—sharing this article with them could help increase their awareness.
The controversial Rothman study concluded that the risk of birth defects was more frequent when women supplemented with high dose vitamin A before the seventh week of gestation. Be aware that some women in this study were likely taking in more than 25,000 IU per day from supplements and/or fortified foods and that these findings have been criticized by other researchers who have found no such correlation. 3,66 Although the fetus is undergoing rapid developmental change during the first trimester of pregnancy, nutrient needs generally do not significantly increase above the nonpregnant state in this early stage.67 Yet, without preformed vitamin A, it is possible for liver stores to become relatively depleted in less than seven months. The World Health Organization recommends vitamin A supplementation of 10,000 IU per day after day sixty of gestation for women who are deficient. Ensuring adequate preformed vitamin A intake in the second or third trimester is beneficial for these reasons, and may reduce complications at birth, especially if a pre-term delivery is anticipated.22
SIDEBARS
QUANTIFYING VITAMIN A
You may be more familiar with quantifying vitamin A in international units, or IU. Supplement labels report vitamin
A amounts in both IU and % daily value and food labels report just the % daily value, which is based on highest IUs needed for an adult. (The current “Nutrition Facts” label on foods will no longer require vitamin A to be listed as of July 2018.) According to FDA policy: 100% of the daily value for vitamin A is 5,000 IU for non-pregnant adults and children four years old or older; in pregnancy the daily value increases to 8,000 IU. The FDA daily values for all nutrients have not changed since 1995, and are based primarily on the 1968 RDAs which were re-established by the IOM starting in 1997. In 2001, the IOM set the current RDA at 770 μg/day RAE for pregnant women 19 years and older, equivalent to 2,570 IU. The IOM set the tolerable upper intake level for all adults 19 years and older at 3,000 μg/day RAE which equates to 10,000 IU of preformed vitamin A. You can arrive at IUs by multiplying RAEs by 3.33.
THE ROTHMAN STUDY
The study usually cited in support of warnings against vitamin A during pregnancy was carried out by KJ Rothman and his team in 1995 at the Boston University School of Medicine and published in The New England Journal of Medicine.1
In the study, researchers asked over twenty-two thousand women to respond to questionnaires about their eating habits and supplement intake before and during pregnancy. Researchers found that cranial-neural crest defects increased with increased dosages of vitamin A; but neural tube defects decreased with increased vitamin A consumption, and no trend was apparent with musculoskeletal, urogenital or other defects.
This study is a poor rack on which to hang the myriad warnings that have kept pregnant women from eating liver
and taking cod liver oil. Researchers made no distinction between synthetic vitamin A derived from multivitamins and processed foods like margarine, and natural vitamin A from food; nor did they take blood samples to determine vitamin A status. Food recall surveys are a notoriously inaccurate method of determining nutrient intake.
Subsequent studies found that high levels of vitamin A did not increase the risk of birth defects. A 1998 study from
Switzerland looked at vitamin A in pregnant women and found that a dose of 30,000 IU per day resulted in blood levels that had no association with birth defects.2
A 1999 study carried out in Rome, Italy found no congenital malformations among one hundred twenty infants
whose mothers consumed an average of 50,000 IU of vitamin A per day.3 Some participants consumed up to 300,000 IU vitamin A daily during pregnancy with no birth defects in the offspring. An average of 50,000 IU vitamin A per day is consistent with WAPF recommendations of cod liver oil to supply 20,000 IU per day plus additional vitamin A in liver, butter, seafood and egg yolks.
REFERENCES
1. Rothman KJ and others. Teratogenicity of high vitamin A intake. N Engl J Med. 1995 Nov 23;333(21):1369-73.
2. Wiegand UW and others. Safety of vitamin A: recent results. Int J Vitam Nutr Res. 1998;68(6):411-6.
3. Holmes LB. Need for inclusion and exclusion criteria for the structural abnormalities recorded in children born from exposed pregnancies. Teratology. 1999 Jan;59(1):1-2.
VITAMIN A SUPPLEMENTATION FOR PREGNANT WOMEN?
The assumption that the vitamin A intakes of pregnant women in the U.S. are more than adequate is widespread.
Between 1981 and 1989, the reported average intakes in studies of pregnant women ranged from 711 to 1440 RE (2,370 to 4800 IU), which exceeded the then RDA of 800 retinol equivalents (RE)1 similar to the current RDA of 770 RAE (although REs were quantified using the more liberal conversion factors for carotenoids to retinol, which have been shown to be overestimated by at least a factor of two). Today, the average dietary intake among females nineteen to thirty years is 515 RAE (1720 IU)2 which may represent a 50 percent decrease from the 1980s, or could reflect the more accurate conversion factor used today. In 2001 the IOM recommended a new unit, the retinol activity equivalent (RAE). Each μg RAE corresponds to 1 μg retinol, 2 μg of β-carotene in oil, 12 μg of “dietary” beta-carotene, or 24 μg of the three other dietary provitamin-A carotenoids.3
In 1990, the Food and Nutrition Board of the IOM recommended “carefully supervised supplementation” for certain
at-risk groups of pregnant women, stressing that “supplementation of vitamin A should be approached with caution
until the risk is clarified.”1 This recommendation came prior to the Rothman study (see sidebar, page 20), which argued against the use of vitamin A by pregnant women, yet there were already some indications that large doses of vitamin A were problematic—chiefly based upon outcomes in women who took the acne medication Isotretinoin during the first trimester. At that time, most scientists suggested that an intake of at least 20,000 to 50,000 IU per day was associated with an increased risk for birth defects.
A few researchers have made efforts to draw attention to this issue. It is “in our opinion rather likely, that a significant portion of the low-income population in some of the most industrialized countries suffers from undiagnosed low vitamin A status. Women in these populations. . . would benefit from a safely designed vitamin A supplementation protocol. However, to our knowledge, there has been no attempt to identify these women or to correct their nutritional deficiencies.”4,5
1. Committee on Nutritional Status During Pregnancy and Lactation, Institute of Medicine, ed. Nutrition During Pregnancy: Part II: Nutrient Supplements. Washington, US: National Academies Press, 1990. http://www.nap.edu/download/1451
2. Scientific Report of the 2015 Dietary Guidelines Advisory Committee. Part E.Section 2.1. Appendix E-2.1: Usual Intake Distributions, 2007-2010, by Age/Gender Groups. http://health.gov/dietaryguidelines/2015-BINDER/meeting2/docs/refMaterials/Usual_Intake_072013.pdf
3. Dietary reference intakes for vitamin A, vitamin K, arsenic, boron, chromium, copper, iodine, iron, manganes(TRUNCATED), 2001, National Academy Press, Washington, D.C. http://www.nap.edu/read/10026/chapter/6#141
4. Azaïs-Braesco V.Pascal G. Vitamin A in pregnancy: requirements and safety limit. Am J Clin Nutr. 2000;71:1325S-1333S.
5. Stephens D, Jackson PL, Gutierrez Y. Subclinical vitamin A deficiency: a potentially unrecognized problem in the United States. Pediatr Nurs 1996;22:377–89.

REFERENCES
1. Azaïs-Braesco V.Pascal G. Vitamin A in pregnancy: requirements and safety limit. Am J Clin Nutr. 2000;71:1325S-1333S.
2. Price WA. Nutrition and Physical Degeneration: 6th Edition. La Mesa, CA: Price-Pottenger Nutrition Foundation (2004) pp. 339-349. – See more at: http://www.westonaprice.org/childrens-health/vitamins-for-fetal-development-conception-to-birth/
3. Masterjohn C. Vitamins for Fetal Development: Conception to Birth. http://www.westonaprice.org/childrens-health/vitamins-for-fetal-development-conception-to-birth/
4. The RDA is the recommended intake needed by nearly all of the population to ensure adequate liver stores of vitamin A in the body (20 μg/g for four months if the person consumes a vitamin A-deficient diet) to support normal reproductive function, immune function, gene expression, and vision. http://lpi.oregonstate.edu/mic/vitamins/vitamin-A#RAE
5. The AI for infants is based on the average amount of retinol supplied by human milk. http://www.nap.edu/read/10026/chapter/6#109
6. Second National Report on Biochemical Indicators of Diet and Nutrition in the U.S. Population, 2012. http://www.cdc.gov/nutritionreport/pdf/Nutrition_Book_complete508_final.pdf
7. Dietary reference intakes for vitamin A, vitamin K, arsenic, boron, chromium, copper, iodine, iron, manganese (TRUNCATED), 2001, National Academy Press, Washington, D.C. http://www.nap.edu/read/10026/chapter/6#141
8. Kemmann E, Pasquale SA, Skaf R. Amenorrhea associated with carotenemia. J Am Med Assoc. 1983;249(7):926-929.
9. Harrison et al. Naturally occurring eccentric cleavage products of provitamin A: β-carotene function as antagonists of retinoic acid receptors. J Biol Chem. 2012;4;287(19):15886-95.
10. Scientific Report of the 2015 Dietary Guidelines Advisory Committee. Part E.Section 2.1. Appendix E-2.1: Usual Intake Distributions, 2007-2010, by Age/Gender Groups. http://health.gov/dietaryguidelines/2015-BINDER/meeting2/docs/refMaterials/Usual_Intake_072013.pdf
11. Scientific Report of the 2015 Dietary Guidelines Advisory Committee.Part E. Section 2. Usual intake distributions, 2007-2010, for pregnant and nonpregnant women in the U.S. ages 19-50 years. http://health.gov/dietaryguidelines/2015-scientific-report/PDFs/Appendix-E-2.4.pdf
12. Intake data on pregnant women is also concerning, with 26% below the EAR of 550 μg, a figure the USDA says is less reliable due to a very small sample size of n=133.
13. Scientific Report of the 2015 Dietary Guidelines Advisory Committee.Part D.Ch 1.Nutrient Intake and Nutrients of Concern. http://health.gov/dietaryguidelines/2015-scientificreport/06-chapter-1/d1-2.asp
14. http://www.fda.gov/Food/GuidanceRegulation/GuidanceDocumentsRegulatoryInformation/DietarySupplements/ucm070620.htm. Last updated 5/27/2015.
15. http://www.fda.gov/Food/GuidanceRegulation/GuidanceDocumentsRegulatoryInformation/LabelingNutrition/ucm385663.htm
16. Second National Report on Biochemical Indicators of Diet and Nutrition in the U.S. population. http://www.cdc.gov/nutritionreport/pdf/Nutrition_Book_complete508_final.pdf
17. Tanumihardjo SA. Biomarkers of vitamin A status: what do they mean? In: World Health Organization. Report: Priorities in the assessment of vitamin A and iron status in populations, Panama City, Panama, 15–17 September 2010. Geneva, World Health Organization, 2012. http://www.who.int/nutrition/publications/micronutrients/background_paper2_report_assessment_vitAandIron_status.pdf
18. World Health Organization. Vitamin A supplementation during pregnancy. Guidance Summary.who.int/elena/titles/guidance_summaries/vitamina_pregnancy/en/
19. Rice AL, et al. Maternal Vitamin A or B-carotene supplementation in lactating Bangladeshi women benefits mothers and infants but does not prevent subclinical deficiency. J Nutr. 1999;129:356-365.
20. Koletzo B, et al. German National Consensus Recommendations on Nutrition and Lifestyle in Pregnancy by the ‘Healthy Start – Young Family Network’. Ann Nutr Metab. 2013;63:311-322.
21. Strobel M, Tinz J, Biesalski H-K. The importance of β-carotene as a source of vitamin A with special regard to pregnant and breastfeeding women. Eur J Nutr. 2007;46:(S1)1-20.
22. Gluckman, Peter, Mark Hanson, Chong Yap Seng, and Anne Bardsley. Nutrition and Lifestyle for Pregnancy and Breastfeeding. Oxford: Oxford UP, 2015.
23. British Nutrition Foundation : Nutrition and Development : Short and Long Term Consequences for Health (1). Somerset, GB: Wiley-Blackwell, 2013. ProQuest ebrary. Web. 26 May 2016.
24. The UK uses plasma retinol as the biochemical marker for their population, stating there is no indication of a prevalence of vitamin A deficiency. Plasma retinol can only detect gross deficiencies. Miller R. Spiro A, Stanner S. Micronutrient status and intake in the UK – where might we be in 10 years time? Nutr Bull. 2016; 41(1):14-41.
25. Masterjohn C. Beyond Good and Evil. http://www.westonaprice.org/health-topics/abcs-of-nutrition/beyond-good-and-evil/
26. USDA Agricultural Research Service National Nutrient Database for Standard Reference Release. https://ndb.nal.usda.gov/ndb/nutrients/report/nutrientsfrm?max=25&offset=0&totCount=0&nutrient1=319&nutrient2=&nutrient3=&subset=0&fg=&sort=c&measureby=m.
27. Common Genetic Variants and Other Host-related factors Greatly Increase Susceptibility to Vitamin A Deficiency. Pizzorno L. 2/12/2016. Longevity Medicine Review.lmreview.com.
28. Hickenbottom et al. Variability in conversion of betacarotene to vitamin A in men as measured by using a double-tracer study design. Am J Clin Nutr. 2002;75:900-907.
29. Lin et al. Variability of the conversion of beta-carotene to vitamin A in women measured by using a double-tracer study design. Am J Clin Nutr. 2000 Jun;71(6):1545-1554.
30. Dietary reference intakes for vitamin A, vitamin K, arsenic, boron, chromium, copper, iodine, iron, manganese
(TRUNCATED), 2001, National Academy Press, Washington, D.C. http://www.nap.edu/read/10026/chapter/6#107
31. The Embryo Project, funded in part by the National Science Foundation. Their open-access Encyclopedia “aims to show readers how to think scientifically—how to identify evidence, how to generate it from experiments, how to relate it to theories, and how to change beliefs and revise theories in light of new evidence.“ https://embryo.asu.edu/pages/retinoids-teratogens#sthash.hOQxe6nK.dpufis
32. Brown B, Mason G, Tang T. Retinoids and pregnancy: an update.Ob & Gyn. 2014;16:7-11. http://onlinelibrary.wiley.com/doi/10.1111/tog.12075/pdf
33. http://www.babycentre.co.uk/x555571/is-it-safe-to-eat-liver-during-pregnancy#ixzz47h9Lp026. My preliminary research indicated that warnings against consumption of rich sources of preformed vitamin A were even more common in the United Kingdom.
34. Razaitis L. The Liver Files. http://www.westonaprice.org/health-topics/the-liver-files/
35. Brown, Judith E. Every Woman’s Guide to Nutrition. Minneapolis: U of Minnesota, 1991.
36. Widdowson, Elsie M., and John C. Mathers. The Contribution of Nutrition to Human and Animal Health. Cambridge: Cambridge UP, 1992.
37. http://www2.erie.gov/health/sites/www2.erie.gov.health/files/uploads/pdfs/he-pam-200.pdf
38. Schoenfeld P. Vitamin B6, The Under-Appreciated Vitamin. http://www.westonaprice.org/health-topics/abcs-of-nutrition/vitamin-b6-the-under-appreciated-vitamin/
39. IOM Dietary Reference Intakes. http://www.nap.edu/read/10026/chapter/6#108
40. http://americanpregnancy.org/pregnancy-health/diet-during-pregnancy/
41. Micozzi M. Inside Vitamin A. Douglas Laboratories Nutri News, May 2009.
42. http://www.marchofdimes.org/pregnancy/foods-to-avoid-or-limit-during-pregnancy.aspx#
43. http://www.mayoclinic.org/healthy-lifestyle/pregnancy-week-by-week/in-depth/pregnancy-nutrition/art-20043844?pg=1
44. Sahin, Nevin Hotun., and Ilkay Gungor. Birth Defects: Issues on Prevention and Promotion. New York: Nova Science, 2010.
45. Cunningham TJ, Duester G. Mechanisms of retinoic acid signalling and its roles in organ and limb development. Nat Rev Mol Cell Biol. 2015;16(2):110-123.
46. Das B et al. Retinoic signalling pathways in development and disease. Bioorg Med Chem. 2014;22(2):673-683.
47. https://chriskresser.com/nourishing-a-growing-baby/
48. Getz KD. Short interpregnancy interval and gastroschisis risk in the National Birth Defects Prevention Study. Birth Defects Res A Clin Mol Teratol. 2012;94(9):714-720.
49. Rothman KJ, Moore LL, Singer MR, Nguyen U-SDT, Mannino S, Milunsky A. Teratogenicity of high vitamin A intake. N Engl J Med. 1995;333:1369-73. http://www.nejm.org/doi/full/10.1056/NEJM199511233332101
50. Oakley GP, Erickson JD. Vitamin A and Birth Defects. Continuing Caution is Needed. N Engl J Med. 1995;333 (21):1414-1415.
51. Committee on Nutritional Status during Pregnancy, ed. Nutrition During Pregnancy. Washington. US: National Academies Press, 1989.
52. http://www.care2.com/greenliving/is-vitamin-a-safe-for-pregnant-women.html
53. http://news.psu.edu/story/332573/2014/10/30/research/researchers-probe-link-between-newborn-health-and-vitamin
54. Chapter in book.p 161-184 Greer, FR. Vitamins. IN: Neonatal Nutrition and Metabolism. Second Edition. Ed. P. Thureen and W. Hay. Cambridge University Press 2006.
55. National Institutes of Health Office of Dietary Supplements. Vitamin A Fact Sheet for Health Professionals. https://ods.od.nih.gov/factsheets/VitaminAHealthProfessional/#en14
56. Hutten MC, Wolfs TGAM, Kramer BW. Can the preterm lung recover from perinatal stress? Mol Cell Pediatr. 2016;3:15.
57. Shah RS et al. Liver stores of vitamin A in human fetuses in relation to gestational age, fetal size and maternal nutritional status. Br J Nutr. 1987;58(2):181-189.
58. http://www.nap.edu/read/10026/chapter/6#94
59. Tanumihardjo SA. Biomarkers of vitamin A status: what do they mean? In: World Health Organization. Report: Priorities in the assessment of vitamin A and iron status in populations, Panama City, Panama, 15–17 September 2010. Geneva, World Health Organization, 2012.
60. Shenai, JP. Vitamin A supplementation in very low birth weight neonates:rational and evidence. Pediatrics.1999;104(6):1369-1375.
61. Murguia-Peniche T. Vitamin D, Vitamin A, maternal-perinatal consideration: old concepts, new insights, new questions. J Ped. 2013:162 (3 Suppl):S26-30.
62. Personal communication with Chris Masterjohn, PhD, via email on 4/15/2016.
63. Common Genetic Variants and Other Host-related factors Greatly Increase Susceptibility to Vitamin A Deficiency. Pizzorno L. 2/12/2016. Longevity Medicine Review. www.lmreview.com.
64. http://www.nap.edu/read/10026/chapter/6#108
65. Sally Fallon Morell, Wise Traditions 2011 Annual Conference
66. Miller RK et al. Periconceptional Vitamin A use: how much is teratogenic? Reprod Toxic. 1998;12(1):75-88.
67. Ritchie LD, King JC. chapter: Nutrient Recommendations and Dietary Guidelines for Pregnant Women. Book: Handbook of Nutrition and Pregnancy. Ed. Lammi-Keefe CJ, Couch SC, Philipson MD.
This article appeared in Wise Traditions in Food, Farming and the Healing Arts, the quarterly journal of the Weston A. Price Foundation, Summer 2016